Absorption or emission of
ultraviolet or visible light by a molecule depends on
electron transitions between molecular orbital energy
levels, just as absorption or emission of electromagnetic
radiation by an atom is determined by electron transitions
between different energy levels in the atom and the
DEs
for those transitions. Molecular spectra follow rules
analogous to the rules for atomic spectra: energy is
absorbed only when the amount of energy provided matches the
difference in energy, DE,
of 2 energy levels. When an electron goes from a higher to a
lower energy state, a photon of definite wavelength and
frequency is emitted. Every atom or molecule has a
characteristic electronic spectrum depending on its
characteristic DEs.
Because of a molecule's greater complexity, we can often
construct a molecule that will give a particular spectrum,
rather than having to just accept the spectra available as
we do with atoms. This possibility arises because of the
interdependence of molecular orbital energy level values for
the molecule, molecular shape, bonding, and distribution of
electron density within the molecule. Energy transitions in
phenolphthalein indicator, plant pigments, and isolated pi
systems will be examined to illustrate this. Molecular orbital theory provides a
model for the way electromagnetic radiation interacts with
molecules. For example, an electron in the pi bonding
molecular orbital (MO) of an alkene can be excited to a pi
antibonding MO. This is described as a
![]() transition.
transition.
 |
|
For an isolated pi bond the energy separation, DE, between the pi bonding and pi antibonding MO's is large; ultraviolet light with its large energy and short wavelength is needed to excite the pi electron. Molecular orbital theory predicts that the energy difference, DE, between levels will decrease if the double bond is conjugated with another double bond. (Conjugated double bonds have one single bond separating them. They are coplanar and pi electrons can move through out the pi system.) Cogjugation exists when series of alternating double and single bond. This means that the molecule has a single bond between the two double bonds. The term "conjugated" is used in chemistry to refer to a series of alternating single and double bonds. The predicted decrease in DE for conjugated structures is also observed in experiments. Here is a general rule that
describes the effect of double bonds conjugation on the
energy absorbed by the pi system. The greater the
number of conjugated multiple bonds in a compound, the
longer the wavelength of the light that the compound will
absorb. This translates into a smaller energy
requirement for the transition from the highest occupied MO
to the lowest unoccupied MO. This idea is illustrated
schematically in the following diagram for alkenes where the
transition is for |
 |
|
Compound name |
Condensed formula |
Wavelength absorbed |
|
Ethene, ethylene |
CH2::CH2 |
171 nm |
|
1,3-butadiene |
CH2::CHCH::CH2 |
217 nm |
|
Trans 1,3,5-hexatriene |
CH2::CHCH::CHCH::CH2 |
274 nm |
|
b-carotene |
see structure below |
425 nm |
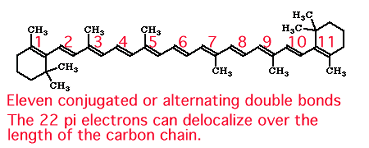
The plant pigment b-carotene has this line structure.
|
There are 11 double bonds in the alternating double bond-single bond system in b-carotene. The double bonds are essentially coplanar and the ¼ electrons are delocalized over a 22 carbon chain. The wavelength absorbed by b-carotene is in the blue region of the visible spectrum. When we look at b-carotene, no "blue wavelengths" reach our eyes because the carotene molecule is absorbing these. The rest of the wavelengths in the visible region (minus the blue) reach our eyes and carotene looks orange to us. Remember, the color of light absorbed by the molecule is not the color we see. It is the one we don't see! |
|
|
|
|
|
Green |
Red |
700 nm |
|
Blue-green |
Orange-red |
600 nm |
|
Violet |
Yellow |
550 nm |
|
Red-violet |
Yellow-green |
530 nm |
|
Red |
Blue-green |
500 nm |
|
Orange |
Blue |
450 nm |
|
Yellow |
Violet |
400 nm |
return to top of page
|
In the first part of this experiment you will observe this difference in color for phenolphthalein and its anion. |
|
In the second part of this experiment you will extract chlorophyll from green leaves and then use chromatography to separate chlorophyll a from chlorophyll b. You may also see the separation of carotene and other plant pigments. You will be asked to explain the different colors of the 2 chlorophylls by examining their structures and identifying the type of change in molecular orbital energy levels caused by the difference in structure. Paper chromatography is a separation technique that anyone who ever spilled coffee or tea on a piece of paper has seen. The solvent wets the paper, creeps along carrying solutes along with it. The different dissolved materials move with the solvent, but at different rates because the paper attracts the solutes differently. You will use this behavior in a systematic way to separate pigments in chlorophyll. You will make an extract of leaf pigments by soaking plant leaves in a mixture of cold acetone and ethanol. The extract appears to be green, but other pigment colors may be masked by the strong green tint. You will use paper chromatography to separate any pigments in the extract. The separation occurs because the pigments have different solubilities in the mobile solvent and different absorption affinities (attractions) to the chromatography paper. The separation process will give a chromatogram. |





